Pulmonary hypertension associated with systemic sclerosis
Pulmonary arterial hypertension (PAH) was considered as the leading cause of morbidity and mortality in systemic sclerosis (SSc) in the last two decades .The patients with SSc and PAH have about 50-60% estimated 3 year survival. There is proliferation and contraction of smooth muscle cells of the pulmonary arterioles in PAH.1
The prevalence of PAH associated with scleroderma is 8-12%.2,3
In patients with diffuse cutaneous SSc (dc SSc) frequency of PAH is almost similar to limited cutaneous SSc.4,5
Mechanisms of pulmonary hypertension in systemic sclerosis:
-PH with proliferative pulmonary vasculopathy = PAH
-PH associated with lung fibrosis due to prominent parenchymal destruction.
– Pulmonary Venous hypertension ( post-capillary PH).
– Post-capillary PH ‘out of proportion’ to that expected from PCWP
– pulmonary veno-occlusive disease(PVOD)
– Porto –pulmonary hypertension (POPH)
-Or any combination of these. 1
PVOD is an important point in systemic sclerosis,therefore briefly explained:
In PVOD vascular lesions are more diffuse.In patients with systemic sclerosis and PVOD,there is also arteriolar microangiopathy and it may causes pulmonary edema after treatment with PAH specific vasodilators.6
HRCT findings include septal lines,lymph node enlargement and centrolobular ground glass opacities.7
In PVOD, the pulmonary vasodilators are usually not useful and sometimes can cause pulmonary edema, therefore the diagnosis of PVOD is critical.
Pulmonary hypertension diagnosis
The symptoms and Clinical examination
The symptoms of PH are non-specific and include dyspnea on exertion, decreasing exercise tolerance, fatigue ,syncope and chest pain.1
The physical signs include a loud P2, left parasternal lift, an RV third heart sound, murmur of tricuspid regurgitation, hepatomegaly and peripheral edema(due to tightness of the skin may not be clear).8
Electrocardiogram (ECG)
Right axis deviation, right ventricle (RV) hypertrophy, P pulmonale,and right bundle branch block are ECG abnormalities in PH.9
Chest radiograph
Findings of chest radiography include central pulmonary arterial dilatation, ‘pruning’ (loss)
of the peripheral branches ,and in more advanced conditions dilatation of right atrium and ventricle.10
Pulmonary function tests and arterial blood gases
These tests reveal the underlying airway or parenchymal lung disease to what extent are involved in development of PH.11,12
Echocardiography
Transthoracic echocardiography is the most effective screening method.
Calculation of pulmonary systolic pressure:
The RV-to right atrium( RA) systolic pressure gradient is calculated from:
ΔP RV-RA=4(VTR peak)²
VTR peak:the maximum velocity in the tricuspid regurgitant(TR) jet.
To determine right ventricular systolic pressure, right atrial pressure(estimated from the size and respiratory variation in the inferior vena cava),is added to ΔP RV-RA.
Pulmonary artery systolic pressure ( s PAP ) and right ventricular systolic pressure are the same if there is not pulmonic valve stenosis.
In summary:
s PAP = ۴ (TRV)² + RA pressure
The assessment of echocardiographic probability of pulmonary hypertension based on tricuspid regurgitation velocity ( TRV) and several additional echocardiographic signs are summarized in tables 1 and 2.8
Table 1 – Echocardiographic probability of pulmonary hypertension in symptomatic patients with a suspicion of pulmonary hypertension
| Peak tricuspid regurgitation
velocity (m/s) |
Presence of other echo
‘PH signs’ a |
Echocardiographic
probability of pulmonary hypertension |
| ≤۲٫۸ or not measurable | No | L Low |
| ≤۲٫۸ or not measurable | Yes | Intermediate |
| ۲٫۹–۳٫۴ | No | Intermediate |
| ۲٫۹–۳٫۴ | Yes | High |
| >3.4 | Not required | High |
PH = pulmonary hypertension
a See next table
Table 2 – Echocardiographic signs suggesting pulmonary hypertension used to assess the probability of pulmonary hypertension in addition to tricuspid regurgitation velocity
| A: The ventricles a | B: Pulmonary artery a | C: Inferior vena cava and right atrium a |
| Right ventricle/ left ventricle basal
diameter ratio >1.0 |
Right ventricular outflow Doppler acceleration time <105 msec and/or
midsystolic notching |
Inferior cava diameter >21 mm with
decreased inspiratory collapse (<50 % with a sniff or <20 % with quiet inspiration) |
| Flattening of the interventricular
septum (left ventricular eccentricity index >1.1 in systole and/or diastole) |
Early diastolic pulmonary regurgitation velocity >2.2 m/sec | Right atrial area (end-systole) >18 cm2 |
| PA diameter >25 mm. |
PA = pulmonary artery.
a Echocardiographic signs from at least two different categories (A/B/C) from the list should be present to alter the level of echocardiographic probability of pulmonary hypertension.
Biomarkers:
Based on Mukerjee et al.,2003 and Williams et al.,2006; N-terminal pro-brain natriuretic peptide(NT-pro BNP) values significantly correlated with haemodynamics.13,2
We can detect an additional 27% of patients with pulmonary hypertension by adding serum NT-pro BNP>100 pg/ml and DLCO<60% of expected to the echocardiographic findings.14
Right heart catheterization(RHC)
To confirm the diagnosis of pulmonary hypertension and to assess the severity of haemodynamic impairment ,RHC is needed.15During RHC, evaluation of the pressure of PA,PA wedge position,RV and RA must be done.Left heart catheterization in addition to RHC should be considered in patients with echocardiographic signs of systolic and/or diastolic LV dysfunction,as well as in patients with risk factors for coronary artery disease or heart failure with preserved ejection fraction. Blood samples should be taken from the high SVC,IVC,PA and systemic artery for oximetry.8
Hemodynamic definitions of pulmonary hypertension:
Based on 2015European Society of Cardiology (ESC) and the European Respiratory Society (ERS) guidelines for the diagnosis and treatment of pulmonary hypertension, hemodynamic definitions of pulmonary hypertension has been shown in table 3.
Table 3 –Haemodynamic definitions of pulmonary hypertension
| Definition | Characteristics |
| PH | PAPm ≥۲۵ mmHg |
| Pre-capillary PH | PAPm ≥۲۵ mmHg
PAWP ≤۱۵ mmHg |
| Post-capillary PH
Isolated post-capillary PH (Ipc-PH) Combined post-capillary and pre-capillary PH (Cpc-PH) |
PAPm ≥۲۵ mmHg
PAWP >15 mmHg DPG <7 mmHg and/or PVR ≤۳ WU DPG ≥۷ mmHg and/or PVR >3 WU |
CO = cardiac output; DPG = diastolic pressure gradient (diastolic PAP – mean PAWP); mPAP = mean pulmonary arterial pressure; PAWP = pulmonary arterial wedge pressure; PH = pulmonary hypertension; PVR = pulmonary vascular resistance; WU = Wood units.
After above evaluations, risk assessment of patients will be performed based on table 4.8
Table 4 – Risk assessment in pulmonary arterial hypertension
| Determinants of prognosisª
(estimated 1-year mortality) |
Low risk <5% | Intermediate risk 5–۱۰% | High risk >10% |
| Clinical signs of right heart failure | Absent | Absent | Present |
| Progression of symptoms | No | Slow | Rapid |
| Syncope | No | Occasional syncopeb | Repeated syncopec |
| WHO functional class | I, II | III | IV |
| NT-proBNP plasma levels | BNP <50 ng/l
NT-proBNP <300 ng/l |
BNP 50–۳۰۰ ng/l
NT-proBNP 300–۱۴۰۰ ng/l |
BNP >300 ng/l
NT-proBNP >1400 ng/l |
| echocardiography | RA area <18 cm2
No pericardial effusion |
RA area 18–۲۶ cm2
No or minimal, pericardial effusion |
RA area >26 cm2
Pericardial effusion |
| Haemodynamics | RAP <8 mmHg CI ≥۲٫۵ l/min/m2 SvO2 >65% | RAP 8–۱۴ mmHg
CI 2.0–۲٫۴ l/min/m2 SvO2 60–۶۵% |
RAP >14 mmHg
CI <2.0 l/min/m2 SvO2 <60% |
BNP = brain natriuretic peptide; CI = cardiac index; NT-proBNP = N-terminal pro-brain natriuretic
peptide; RA = right atrium; RAP = right atrial pressure; SvO2 = mixed venous oxygen saturation; WHO =World Health Organization.
ª Most of the proposed variables and cut-off values are based on expert opinion. They may provide prognostic information and may be used to guide therapeutic decisions, but application to individual patients must be done carefully. One must also note that most of these variables have been validated mostly for IPAH and the cut-off levels used above may not necessarily apply to other forms of PAH. Furthermore, the use of approved therapies and their influence on the variables should be considered in the evaluation of the risk.
b Occasional syncope during brisk or heavy exercise, or occasional orthostatic syncope in an otherwise stable patient.
c Repeated episodes of syncope, even with little or regular physical activity.
In asymptomatic patients with systemic sclerosis,transthoracic echocardiography is recommended as a screening test.After this,annual screening with echocardiography,DLCO and biomarkers is recommended.8,16,17
In our center ,decision making for the next step after the echocardiography is based on the table 5.
Table 5 – Diagnostic management suggested according to echocardiographic probability of pulmonary hypertension
| Echocardiographic probability of PH | Next step in management |
| Low | Echo follow-up should be considered |
| Intermediate | Further assessment of PH including RHC should be considered |
| High | RHC |
If the patient refuses to RHC,after twice echocardiography within three months with evidence of intermediate to high echocardiographic probability of pulmonary hypertension and after R/O PVOD, we start the treatment of PH with Phosphodiesterase type5 inhibitors (Sildenafil or Tadalafil) due to the mechanism of action of these drugs and vascular problem in these patients.
Therapy
Based on 2015 ESC and ERS guidelines for the diagnosis and treatment of pulmonary hypertension, the overall treatment goal in patients with PAH is achieving a low risk status (Table 4), “which is usually associated with good exercise capacity, good quality of life, good RV function and a low mortality risk”.
General measures
The patients should be encouraged to be active to tolerable extent but should avoid excessive physical activities.
The 2015 PH guidelines suggested that oral anticoagulation may be considered on an individual basis and in presence of thrombophilic predisposition. Class IIb, Level c.8,18,19
Based on the 2015 PH guidelines, diuretic treatment is recommended in patients with signs of RV failure and fluid retention. Class I, Level c.8,20
Continuous long-term O2 therapy is recommended in patients when arterial blood O2 pressure is consistently < 60 mmHg. Class I, Level c.8,21
Iron deficiency has been reported in 46% of patients with SSc-PAH.22
Therefore regular monitoring of the iron status should be considered in these patients. If iron deficiency is detected, a search for potential causes must be done and treatment should be considered.8
Specific drug therapy
Endothelin receptor antagonists
Bosentan , an oral active dual endothelin receptor type A and B antagonist, of this class is recommended.
The starting dosage is 62.5 mg twice daily and increased to 125 mg twice daily after 1month if there are no liver function test derangements or other important adverse effects.1
In patients receiving bosentan, liver function testing should be performed monthly.8
Phosphodiesterase type 5 inhibitors
Sildenafil and tadalafil are recommended of this class. Sildenafil,20 mg three times daily and tadalafil,20 mg once daily(and then increase to 40 mg daily) are recommended.8
Most side effects of these drugs are mainly related to vasodilation and including headache, flushing and epistaxis.1,23
Prostacyclin analogues and prostacyclin receptor agonists
Iloprost is a chemically stable prostacyclin analogue available as intravenous or aerosol administration.
Inhaled iloprost
Overall, inhaled iloprost was well tolerated. The inhaled form has been shown to improve exercise tolerance and symptoms when administered 6-9 times daily.1 The most frequent side effects are jaw pain and flushing .
Intravenous iloprost
Intravenous iloprost with starting dose of 0.5ng/kg/min and increase to maximum dose 2ng/kg/min for 6 hour in 5 consecutive days, has shown exercise performance and hemodynamic benefits.1
Transplantation
SSc should not be considered as an a priori contraindication for lung transplantation.8,24
About indications and contraindications for transplantation in patients with systemic sclerosis, decision making must be with a special focus on digestive (gastro-esophageal reflux disease and intestinal disease), cardiac, renal and cutaneous involvement. 8
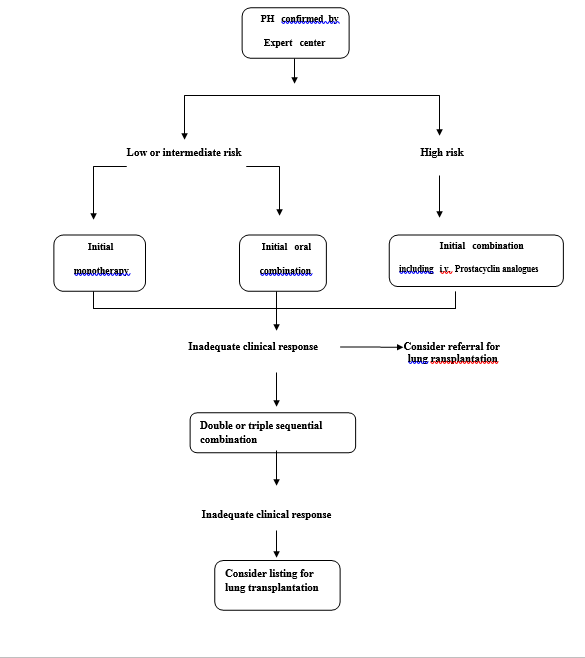
The treatment algorithm of our center is shown in figure 1.Achievement/maintenance of a low-risk profile (Table 4) considered to be an adequate treatment response .
Figure 1 -Treatment algorithm for pulmonary arterial hypertension
In our center assessment of the patients and timing for the follow-up is based on table7.
Table 6 –Suggested assessment and timing for the follow-up of patients
| At baseline | Every 3–۶
monthsa |
Every 6–۱۲
monthsa |
۳–۶ months after changes in therapya | In case of clinical
worsening |
|
| Medical assessment and
determination of functional class |
+ | + | + | + | + |
| ECG | + | + | + | + | + |
| Echo | + | + | + | + | |
| Right heart catheterization | + | + | + |
a Intervals to be adjusted according to patient needs.
References
۱٫Hachulla, E.,Coghlan ,G., EULAR Textbook on Systemic Sclerosis,ch.22 ( Pulmonary arterial hypertension associated with scleroderma) ,2013.
۲٫Mukerjee, D., St George, D., Coleiro, B., Knight, C., Denton, C.P., Davar, J., Black, C.M. and Coghlan, J.G., 2003. Prevalence and outcome in systemic sclerosis associated pulmonary arterial hypertension: application of a registry approach. Annals of the rheumatic diseases, 62(11), pp.1088-1093.
۳٫Hachulla, E., Launay, D., Mouthon, L., Sitbon, O., Berezne, A., Guillevin, L., Hatron, P.Y., Simonneau, G., Clerson, P. and Humbert, M., 2009. Is pulmonary arterial hypertension really a late complication of systemic sclerosis?. CHEST Journal, 136(5), pp.1211-1219.
۴٫Hunzelmann, N., Genth, E., Krieg, T., Lehmacher, W., Melchers, I., Meurer, M., Moinzadeh, P., Müller-Ladner, U., Pfeiffer, C., Riemekasten, G. and Schulze-Lohoff, E., 2008. The registry of the German Network for Systemic Scleroderma: frequency of disease subsets and patterns of organ involvement. Rheumatology, 47(8), pp.1185-1192.
۵٫Nihtyanova, S.I., Tang, E.C., Coghlan, J.G., Wells, A.U., Black, C.M. and Denton, C.P., 2010. Improved survival in systemic sclerosis is associated with better ascertainment of internal organ disease: a retrospective cohort study. Qjm, 103(2), pp.109-115.
۶٫Montani, D., Achouh, L., Dorfmüller, P., Le Pavec, J., Sztrymf, B., Tchérakian, C., Rabiller, A., Haque, R., Sitbon, O., Jaïs, X. and Dartevelle, P., 2008. Pulmonary veno-occlusive disease: clinical, functional, radiologic, and hemodynamic characteristics and outcome of 24 cases confirmed by histology. Medicine, 87(4), pp.220-233.
۷٫Günther, S., Jaïs, X., Maitre, S., Berezne, A., Dorfmüller, P., Seferian, A., Savale, L., Mercier, O., Fadel, E., Sitbon, O. and Mouthon, L., 2012. Computed tomography findings of pulmonary venoocclusive disease in scleroderma patients presenting with precapillary pulmonary hypertension. Arthritis & Rheumatism, 64(9), pp.2995-3005.
۸٫۲۰۱۵ ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension ,The Joint Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS)
- Bonderman D, Wexberg P, Martischnig AM, Heinzl H, Lang MB, Sadushi R,Skoro-Sajer N, Lang IM. A noninvasive algorithm to exclude pre-capillary pulmonary hypertension. Eur Respir J 2011;37:1096–۱۱۰۳٫
- Milne EN. Forgotten gold in diagnosing pulmonary hypertension: the plain chest radiograph. Radiographics 2012;32:1085–۱۰۸۷٫
- Trip P, Nossent EJ, de Man FS, van den Berk IA, Boonstra A, Groepenhoff H, Leter EM,Westerhof N, Grunberg K, Bogaard HJ, Vonk-Noordegraaf A. Severely reduced diffusion capacity in idiopathic pulmonary arterial hypertension: patient characteristics and treatment responses. Eur Respir J 2013;42:1575–۱۵۸۵٫
- Sun XG, Hansen JE, Oudiz RJ,Wasserman K. Pulmonary function in primary pulmonary hypertension. J Am Coll Cardiol 2003;41:1028–۱۰۳۵٫
۱۳٫Williams, M.H., Das, C.L.A.R.E., Handler, C.E., Akram, M.R., Davar, J.O.S.E.P.H., Denton, C.P., Smith, C.J., Black, C.M. and Coghlan, J.G., 2006. Systemic sclerosis associated pulmonary hypertension: improved survival in the current era. Heart, 92(7), pp.926-932.
۱۴٫Khanna,D.,Saggar,R.,Furst,D.E.,et al.( 2010) Predictive value of non-invasive tests for the diagnosis of scleroderma-associated pulmonary hypertension: PHAROS registry . [abstract].Arthritis and Rheumatism,62(Suppl 10),p.729.
- Hoeper MM, Lee SH, Voswinckel R, Palazzini M, Jaı¨s X, Marinelli A, Barst RJ,
Ghofrani HA, Jing ZC, Opitz C, Seyfarth HJ, Halank M, McLaughlin V, Oudiz RJ,
Ewert R, Wilkens H, Kluge S, Bremer HC, Baroke E, Rubin LJ. Complications of
right heart catheterization procedures in patients with pulmonary hypertension
in experienced centers. J Am Coll Cardiol 2006;48:2546–۲۵۵۲٫
- Hachulla E, Gressin V, Guillevin L, Carpentier P, Diot E, Sibilia J, Kahan A, Cabane J,
Frances C, Launay D, Mouthon L, Allanore Y, Kiet PT, Clerson P, de Groote P,Humbert M. Early detection of pulmonary arterial hypertension in systemic sclerosis: a French nationwide prospective multicenter study. Arthritis Rheum 2005;52: 3792–۳۸۰۰٫
- Khanna D, Gladue H, Channick R, Chung L, Distler O, Furst DE, Hachulla E, Humbert M, Langleben D, Mathai SC, Saggar R, Visovatti S, Altorok N,Townsend W, FitzGerald J, McLaughlin VV. Recommendations for screening and detection of connective tissue disease-associated pulmonary arterial hypertension. Arthritis Rheum 2013;65:3194–۳۲۰۱٫
- Olsson KM, Delcroix M, Ghofrani HA, Tiede H, Huscher D, Speich R, Gru¨nig E,
Staehler G, Rosenkranz S, Halank M, Held M, Lange TJ, Behr J, Klose H,
Claussen M, Ewert R, Opitz CF, Vizza CD, Scelsi L, Vonk-Noordegraaf A,
Kaemmerer H, Gibbs JS, Coghlan G, Pepke-Zaba J, Schulz U, Gorenflo M,
Pittrow D, Hoeper MM. Anticoagulation and survival in pulmonary arterial hypertension:
results from the Comparative, Prospective Registry of Newly Initiated
ESC/ERS Guidelines 117 Downloaded from http://eurheartj.oxfordjournals.org/ by guest on January 22, 2016 Therapies for Pulmonary Hypertension (COMPERA). Circulation 2014;129:
۵۷–۶۵٫
- Jaı¨s X, Launay D, Yaici A, Le PJ, Tcherakian C, Sitbon O, Simonneau G,
Humbert M. Immunosuppressive therapy in lupus- and mixed connective tissue
disease-associated pulmonary arterial hypertension: a retrospective analysis of
twenty-three cases. Arthritis Rheum 2008;58:521–۵۳۱٫
- Cohn JN. Optimal diuretic therapy for heart failure. Am J Med 2001;111:577.
- ۲ Sandoval J, Aguirre JS, Pulido T, Martinez-Guerra ML, Santos E, Alvarado P,
Rosas M, Bautista E. Nocturnal oxygen therapy in patients with the Eisenmenger
syndrome. Am J Respir Crit Care Med 2001;164:1682–۱۶۸۷٫
- ۲ Ruiter G, Lanser IJ, de Man FS, Van Der Laarse WJ, Wharton J, Wilkins MR,
Howard LS, Vonk-Noordegraaf A, Voskuyl AE. Iron deficiency in systemic sclerosis
patients with and without pulmonary hypertension. Rheumatology (Oxford)
۲۰۱۴;۵۳:۲۸۵–۲۹۲٫
- Vachiery JL, Huez S, Gillies H, Layton G, Hayashi N, Gao X, Naeije R. Safety, tolerability
and pharmacokinetics of an intravenous bolus of sildenafil in patients with
pulmonary arterial hypertension. Br J Clin Pharmacol 2011;71:289–۲۹۲٫
۲۴٫ Launay D, Savale L, Berezne A, Le PJ, Hachulla E, Mouthon L, Sitbon O, Lambert B,Gaudric M, Jaı¨s X, Stephan F, Hatron PY, Lamblin N, Vignaux O, Cottin V, Farge D,Wallaert B, Guillevin L, Simonneau G, Mercier O, Fadel E, Dartevelle P,Humbert M, Mussot S. Lung and heart-lung transplantation for systemic sclerosispatients. A monocentric experience of 13 patients, review of the literature and position paper of a multidisciplinary working group. Presse Med 2014;43: e345–e363.